【论著】| 普列克底物蛋白2/miR-196a信号轴介导肿瘤微环境中肺癌细胞的通讯机制研究
时间:2024-08-18 06:00:29 热度:37.1℃ 作者:网络
[摘要] 背景与目的:阐明介导肿瘤相关成纤维细胞(cancer-associated fibroblasts,CAFs)与肿瘤细胞之间通讯的信号分子仍然是一个巨大的挑战,这些信号分子对癌症转移至关重要。本研究旨在探讨普列克底物蛋白2(pleckstrin-2,PLEK2) /miR-196a信号轴介导肿瘤微环境中肺癌细胞的通讯机制。方法:选择人肺腺癌细胞系H1299和人胚胎肺细胞MRC-5作为研究对象。用表达PLEK2的慢病毒(PLEK2)和载体对照(Vector)转染H1299细胞,并在转染24 h后分离外泌体(Vector_exo、PLEK2_exo)。采用miR-196a模拟物或抑制剂转染MRC-5细胞。通过蛋白质印迹法(Western blot)分析PLEK2和上皮-间充质转化(epithelial-mesenchymal transition,EMT)相关蛋白水平,采用聚合酶链反应(polymerase chain reaction,PCR)分析miR-196a表达,采用transwell实验测定细胞转移和侵袭能力。将6只雌性BALB/c-nu小鼠随机分为Vector组和PLEK2组,每组3只。通过尾静脉向各组小鼠注射转染Vector或PLEK2的H1299细胞。4周后,取出肺组织进行H-E染色和免疫组织化学染色分析α-平滑肌肌动蛋白(α-smooth muscle actin,α-SMA)表达。所有动物实验均经长沙市第一医院(中南大学湘雅医学院附属长沙医院)伦理委员会批准(伦理编号为EI-2021-103)。结果:与Vector组相比,PLEK2组小鼠肺结节数和转移灶中α-SMA表达显著增加(P<0.001)。与Vector组相比,PLEK2组H1229细胞中miR-196a表达水平显著增加(P<0.05),并且PLEK2_exo中miR-196a表达水平显著高于Vector_exo(P<0.05)。与Vector_exo组相比,PLEK2_exo组MRC-5细胞中miR-196a、α-SMA和成纤维细胞活化蛋白(fibroblast activation protein,FAP)表达水平显著增加(P<0.05)。与阴性对照(negative control,NC)相比,miR-196a转染的MRC-5细胞中α-SMA和FAP表达水平显著增加(P<0.05)。相反,通过miR-196a抑制剂(si-miR-196a#1、si-miR-196a#)转染,α-SMA和FAP表达水平被显著抑制(P<0.05)。与NC-CM组相比,miR-196a-CM组H1299细胞的转移、侵袭细胞数和波形蛋白(vimentin)表达均显著增加(P<0.001),E-钙黏蛋白(E-cadherin)表达显著降低(P<0.001)。此外,与Vector_exo-CM组相比,PLEK2_exo-CM组H1299细胞的转移、侵袭细胞数和vimentin表达均显著增加(P<0.01),E-cadherin表达显著降低(P<0.001)。结论:PLEK2上调能够增强肺癌细胞来源的外泌体miR-196a水平,从而促进CAFs激活。激活的CAFs能够进一步增强肺癌细胞的侵袭能力。
[关键词] 普列克底物蛋白2;肿瘤微环境;肺癌细胞;肿瘤相关成纤维细胞
[Abstract] Background and purpose: It is still a great challenge to clarify the signal molecules that mediate the communication between cancer-associated fibroblasts (CAFs) and tumor cells. These signal molecules are very important for cancer metastasis. The purpose of this study was to explore the communication mechanism of pleckstrin-2/miR-196a signal axis mediated by lung cancer cells in tumor microenvironment. Methods: Human lung adenocarcinoma cell line H1299 and human embryonic lung cell MRC-5 were selected as the research objects. H1299 cells were transfected with lentivirus (PLEK2) expressing PLEK2 and Vector control, and exosomes (Vector_exo, PLEK2_exo) were isolated after 24 h of transfection. MRC-5 cells were transfected with miR-196a mimetic or inhibitor. The expressions of PLEK2 and epithelial-mesenchymal transition (EMT)-related proteins were analyzed by Western blot. The expression of miR-196a was analyzed by polymerase chain reaction (PCR), and the metastasis and invasion ability of cells were determined by transwell assay. Six female BALB/c-nu mice were randomly divided into Vector group and PLEK2 group, with 3 mice in each group. Mice in each group were injected with H1299 cells transfected with Vector or PLEK2 through the tail vein. After 4 weeks, lung tissue was taken out for H-E staining and immunohistochemical staining to analyze the expression of α-smooth muscle actin (α-SMA). All animal experiments were approved by the ethics committee of First Hospital of Changsha City (Changsha Hospital, Xiangya School of Medicine, Central South University) (ethics number: EI-2021-103). Results: Compared with the Vector group, the number of pulmonary metastatic nodules and the expression of α-SMA in metastatic cancer in PLEK2 group increased significantly (P<0.001). Compared with Vector group, the expression level of miR-196a in H1229 cells in PLEK2 group increased significantly (P<0.05), and the expression level of miR-196a was significantly higher in PLEK2_exo than in Vector_exo (P<0.05). Compared with Vector_exo group, the expression levels of miR-196a, α-SMA and fibroblast activation protein (FAP) in MRC-5 cells in PLEK2_exo group increased significantly (P<0.05). Compared with the negative control (NC), the expression levels of α-SMA and FAP in MRC-5 cells transfected with miR-196a increased significantly (P<0.05). On the contrary, by transfection with miR-196a inhibitors (si-miR-196a#1 and si-miR-196a#), the expression levels of α-SMA and FAP were significantly inhibited (P<0.05). Compared with NC-CM group, the number of metastatic cells, invasive cells and the expression of vimentin in miR-196a-CM group increased significantly (P<0.001), and the expression of E-cadherin decreased significantly (P<0.001). In addition, compared with Vector_exo-CM group, PLEK2_exo-CM group had significant increase in number of metastatic and invasive cells and the expression of vimentin (P<0.01), and significant decrease in the expression of E-cadherin (P<0.001). Conclusion: Upregulation of PLEK2 can enhance the level of exosomes miR-196a derived from lung cancer cells, thereby promoting the activation of CAFs. The activated CAFs can further enhance the invasive ability of lung cancer cells.
[Key words] Pleckstrin-2; Tumor microenvironment; Lung cancer cells; Cancer-associated fibroblasts
肺癌是全球癌症相关死亡的主要原因[1]。尽管已经开发了针对肺癌的多种疗法,但肺癌患者的5年生存率仍远低于其他癌症[2]。肺癌患者预后不良的主要原因是肺癌转移率高[3]。癌细胞的转移潜力取决于它们与多种类型的周围细胞的直接或间接相互作用,如免疫细胞、血管细胞和肿瘤相关成纤维细胞(cancer-associated fibroblasts,CAFs),这些细胞与分泌信号因子和细胞外基质(extracellular matrix,ECM)共同构成肿瘤微环境(tumor microenvironment,TME)[4]。为了提高疗效和患者生存率,迫切需要针对癌细胞和TME的新型抗癌策略。普列克底物蛋白2(Pleckstrin-2,PLEK2)是Pleckstrin家族的成员。目前在肺癌、骨肉瘤和结直肠癌中,关于PLEK2的研究主要集中在肿瘤细胞迁移和侵袭上,并且PLEK2可以作为潜在的治疗靶点[5]。最近一项基于癌症基因组图谱(The Cancer Genome Atlas,TCGA)数据库的生物信息学分析[6]证实,PLEK2是非小细胞肺癌TME的相关基因,并与CAFs激活密切相关。然而, PLEK2在TME中的作用尚未通过实验证明。CAFs是TME中的主要基质细胞,在肿瘤的发展和转移中发挥重要作用。CAFs主要来源于组织常驻成纤维细胞,这些成纤维细胞响应来源于癌细胞和TME的信号而被激活。CAFs通过ECM的重塑促进恶性细胞的生长和迁移[7]。
外泌体(exosomal,exo)是一类直径为50~150 nm的细胞外囊泡,通过运输介质(如蛋白质、脂质和核酸)介导肿瘤和CAFs之间的双向交流[8]。癌细胞来源的外泌体miRNA已被证明在培养基质细胞中起主导作用,特别是将成纤维细胞转化为CAFs,这反过来有助于许多肿瘤的转移[9]。因此,CAFs已被用作抑制转移的治疗靶点。然而,阐明介导CAFs与肿瘤细胞之间通讯的信号分子仍然是一个巨大的挑战,这些信号分子对癌症转移至关重要。本研究旨在探讨PLEK2激活对肺癌细胞中外泌体miRNA释放的影响,以及其在CAFs肺癌细胞之间通讯中的作用。
1 材料和方法
1.1 材料
1.1.1 试剂
RPMI-1640培养基和DMEM培养基均购自美国Gibco公司,LipofectamineTM2000转染试剂购自美国Invitrogen公司,PKH26膜染料购自美国Sigma-Aldrich公司,苯甲基磺酰氟(phenylmethylsulfonyl fluoride,PMSF)和完全蛋白酶抑制剂混合物购自瑞士Roche公司, RIPA缓冲液购自上海碧云天生物技术股份有限公司,二辛可宁酸(bicinchoninic acid,BCA)蛋白质分析试剂盒购自美国Thermo Scientific公司,聚偏二氟乙烯(polyvinylidene fluoride,PVDF)膜购自美国Millipore公司,CD9、 CD81、CD63和钙联蛋白(calnexin)均购自美国Santa Cruz Biotechnology公司,PLEK2、成纤维细胞活化蛋白(fibroblast activation protein,FAP)、钙黏蛋白E(E-cadherin)和波形蛋白(vimentin)均购自英国Abcam公司,化学发光试剂盒购自美国Millipore公司,24孔板transwell小室购自美国Corning公司,抗α-平滑肌肌动蛋白(α-smooth muscle actin,α-SMA)抗体购自英国Abcam公司,mirVana miRNA分离试剂盒购自美国Invitrogen公司,miScript试剂盒和miScript SYBR Green聚合酶链反应(polymerase chain reaction,PCR)试剂盒均购自美国Qiagen公司。
1.1.2 仪器
光学显微镜购自日本Olympus公司,离心机购自美国Beckman Coulter公司,JEM-2100透射电子显微镜购自日本电子株式会社,TCS SP5共聚焦显微镜购自德国Leica公司。
1.1.3 细胞系
人肺腺癌(lung adenocarcinoma,LUAD)细胞系H1299购自美国典型培养物保藏中心,并在补充有10%胎牛血清(fetal bovine serum,FBS)的RPMI-1640培养基中培养。人胚胎肺细胞MRC-5购自江苏凯基生物技术股份有限公司,并在补充有10%FBS的DMEM培养基中培养。所有细胞都在37 ℃、CO2体积分数为5%的潮湿环境中温育。
1.1.4 动物
6只无特定病原体(specific pathogen free,SPF)级4周龄雌性BALB/c-nu小鼠购自上海斯莱克实验动物有限责任公司。所有小鼠饲养在无病原体环境中。
1.2 细胞转染
用表达PLEK2和荧光素酶的慢病毒(PLEK2)和载体对照[Vector,汉恒生物科技(上海)有限公司]转染H1299细胞。为确定miR-196a的作用,使用LipofectamineTM2000转染试剂将100 nmol/L miR-196a模拟物(正义链:5‘-GAGGCAGAAGCAGGAUGACA-3‘;反义链:5‘-UCAUCCUGCUUCUGCCUCUU-3‘)或抑制剂(si-miR-196a#1和si-miR-196a#2,序列分别为5‘-CGCUCUCCUUUACAUU-3‘和5‘-GCCGUUCCCUUCCAUUUAUC-3‘,上海吉玛制药技术有限公司)转染至MRC-5细胞。空载体[阴性对照(negative control,NC),正义链:5‘-UUCUCCGAACGUGUCACGUTT-3‘;反义链:5‘-ACGUGACACGUUCGGAGAATT-3‘]和si-NC(序列为5‘-CAGUACUUUUGUGUA GUACAA-3‘)作为对照。转染48 h后,收集细胞用于进一步实验。
1.3 体内成瘤率测定
小鼠适应性喂养1周后随机分为Vector组和PLEK2组,每组3只。通过尾静脉向各组小鼠注射3×106个转染Vector或PLEK2的H1299细胞。4周后,处死小鼠,取出肺组织标本用4%甲醛溶液固定,石蜡包埋,H-E染色。所有动物实验均经长沙市第一医院(中南大学湘雅医学院附属长沙医院)伦理委员会批准(伦理编号为EI-2021-103)。
1.4 免疫组织化学染色
对所有石蜡包埋组织切片进行脱蜡和再水化。阻断后,将载玻片与抗α-平滑肌肌动蛋白(α-smooth muscle actin,α-SMA)抗体在4 ℃下温育过夜。第2天,用磷酸缓冲盐溶液(phosphate-buffered saline,PBS)洗涤后,将切片与HRP缀合的第二抗体在37 ℃下温育1 h。切片用3,3’-二氨基联苯(3,3’-diaminobenzidine,DAB)溶液染色3 min,并用苏木精复染。使用光学显微镜获取图像。
1.5 生物信息学分析
基因表达谱交互分析(gene expression profile interaction analysis,GEPIA,http://GEPIA. cancer-pku.cn/index.html)用于分析PLEK2表达与α-SMA或FAP的相关性以及PLEK2在肺癌患者不同病理学阶段的表达。使用肺癌的TCGA数据集,通过GEPIA探讨PLEK2表达与总生存期的关系。
1.6 外泌体的分离和鉴定
对于外泌体分离,将Vector或PLEK2转染的H1299细胞用PBS洗涤2次,并加入新鲜的不含FBS的RPMI-1640培养基。24 h后收集培养基通过100 000×g超速离心70 min以分离外泌体(Vector_exo和PLEK2_exo)。
使用JEM-2100透射电子显微镜观察外泌体的形态和大小。通过测定外泌体的特异性标志物CD9、CD81和CD63的表达来鉴定外泌体,将细胞裂解物中的内质网标志物calnexin作为NC。
1.7 外泌体标记和追踪
使用PKH26膜染料对外泌体进行荧光标记,然后与受体细胞MRC-5温育48 h。通过TCS SP5共聚焦显微镜观察外泌体的摄取情况。
1.8 miRNA测序
使用mirVana miRNA分离试剂盒从用Vector或PLEK2转染的H1299细胞和外泌体(Vector_exo、PLEK2_exo)中提取总RNA。由武汉华大基因科技有限公司在BGISEQ-500平台上进行细胞miRNA和外泌体miRNA测序。使用SOAPnuke软件过滤原始读数,并使用HISAT软件将其映射到人类参考基因组GRCh38中。
1.9 反转录PCR(reverse transcription-PCR,RT-PCR)分析
分别使用miScript试剂盒和miScript SYBR Green PCR试剂盒对细胞进行RT-PCR和实时荧光定量PCR(real-time fluorescence quantitative PCR,RTFQ-PCR)分析。由生工生物工程(上海)股份有限公司合成miR-196a、RNU6B、α-SMA、FAP和GAPDH的引物。GAPDH的正义链:5’-CCTCTGACTTCAACAGCGAC-3’;反义链:5’-TCCTTTGTGCTCTTGCTGGC-3’。α-SMA的正义链:5’-CATCACCAACTGGGACGACATGGAA-3’;反义链:5’-GCATAGCCCTCATAGATGGGGA CATTG-3 ’。FAP的正义链:5’-TGCCACC TCTGCTGTGC-3’;反义链:5’-GAAGCATTCAC ACTTTTCATGGT-3 ’。RNU6B的正义链:5’-CTCGCTTCGGCAGCACAT-3’;反义链:5’-AACGCTTCACGAATTTGCGT-3’。miR-196a的正义链:5’-CCGATGTAGGGCTAAAAGCC-3’;反义链:5’-CTCAACTGGTGTCGTGGAGTC-3’。实验重复3次。
1.10 蛋白质印迹法(Western blot)
细胞或外泌体在含有1 mmol/L PMSF和完全蛋白酶抑制剂混合物的冰上RIPA缓冲液中裂解。使用BCA蛋白质分析试剂盒对总蛋白质进行定量。将等量的蛋白质裂解物加载到十二烷基硫酸钠聚丙烯酰胺凝胶电泳(sodium dodecylsulphate polyacrylamide gel electrophoresis,SDS-PAGE)凝胶上,并转移到PVDF膜上。将膜封闭并在4 ℃下与针对CD9、CD81、CD63、calnexin、PLEK2、FAP、E-cadherin和vimentin的初级抗体一起温育过夜。通过化学发光使免疫反应性产物可视化,并使用AlphaView软件通过光密度测定法定量。用GAPDH作为加载对照,对条带进行光密度分析。实验重复3次。
1.11 Transwell实验分析
在侵袭试验中,将小室插入24孔板中,并用基质胶包被至少3 h,以模拟血管基底膜。在迁移试验中,将小室插入没有基质胶涂层的24孔板中。将H1299悬浮在无血清培养基中,并接种到具有8 μm孔径插入物的transwell小室中。将用100 nmol/L miR-196a模拟物和对照转染的MRC-5细胞的条件培养基(miR-196a-CM和NC-CM)或用Vector_exo、PLEK2_exo处理MRC-5细胞的条件培养基(Vector_exo-CM、PLEK2_exo-CM)加入到底部小室中。24 h后,用0.2%结晶紫染液对迁移穿过膜并黏附在膜下表面的细胞进行染色,并在光学显微镜下观察并分析迁移和侵袭细胞数。实验重复3次。
1.12 统计学处理
使用SPSS 20.0软件对数据进行统计学分析。数据以x±s表示。两组间的平均值比较采用t检验。多组间的比较采用单因素方差分析(ANOVA),多重比较采用LSD-t检验。使用DESeq2确定差异表达基因(倍数变化≥2,P <0.05)。使用PLEK2中位数截断值将患者分为高和低基因表达水平进行生存分析,并显示log-rank检验的P值。P<0.05为差异有统计学意义。
2 结 果
2.1 肺癌中PLEK2的高表达与CAFs相关
GEPIA结果表明,LUAD和肺鳞状细胞癌(lung squamous cell carcinoma,LUSC)中PLEK2的表达显著高于正常组织(P<0.05),并且PLEK2的表达随肿瘤分期增加而增加(F=4.73,P<0.05,图1A、1B)。PLEK2的表达与FAP和α-SMA的表达呈显著正相关(R=0.24和0.15,图1C、1D)。此外,PLEK2的高表达与肺癌患者的不良总生存期显著相关(log-rank P<0.05,图1E)。上述结果提示PLEK2的高表达可能与CAFs的激活有关,且能促进肺癌的转移。
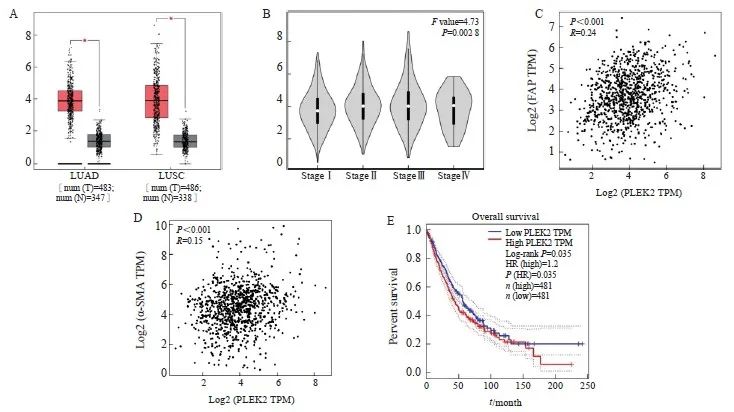
图1 基于GEPIA的PLEK2表达与CAFs相关
Fig. 1 The expression of plek2 is related to CAFs based on GEPIA
A: PLEK2 expression is present in LUAD (including 483 tumor samples and 347 normal samples) and LUSC (including 486 tumor samples and 338 normal samples). The red box chart represents tumor tissue, while the gray box chart represents normal tissue. The vertical axis represents the logarithmic value of PLEK2 per million transcripts [log2 (PLEK2 TPM)]. B: The relative expression of PLEK2 in different pathological stages of lung cancer (LUAD+LUSC) showed significant differences (F=4.73, P=0.002 8). The vertical axis represents the logarithmic value of the copy number of the PLEK2 transcript [log2 (PLEK2 TPM)]. C: The expression of PLEK2 (log2 PLEK2 TPM) in lung cancer (LUAD+LUSC) was positively correlated with the expression of α-SMA (log2-SMA TPM) (R=0.24, P<0.001). D: The expression of PLEK2 (log2 PLEK2 TPM) in lung cancer (LUAD+LUSC) was positively correlated with the expression of FAP (log2 FAP TPM) (R=0.15, P<0.001). TPM stands for every million transcripts. E: Using the TCGA data set of lung cancer patients (LUAD+LUSC), the correlation between the expression of PLEK2 and the overall survival was analyzed by GEPIA. Patients were divided into high (red,n=481) and low (blue, n=481) gene expression levels by using the median cut-off value, and the log-rank P=0.035 was displayed, indicating that the difference was statistically significant.
2.2 PLEK2过表达促进肺癌细胞的上皮-间充质转化(epithelial-mesenchymal transition,EMT)
通过Western blot检测H1299细胞的EMT相关蛋白表达,与Vector组相比,PLEK2组H1299细胞中E-cadherin表达显著下调,vimentin表达显著上调(P<0.05),表明PLEK2上调促进H1299细胞的EMT(图2A)。动物实验直接通过静脉注射肺癌细胞以观察肺中的成瘤率,与Vector组相比,PLEK2组小鼠肺形成的结节数和转移癌中α-SMA表达显著增加(P<0.001,图2B、2C)。
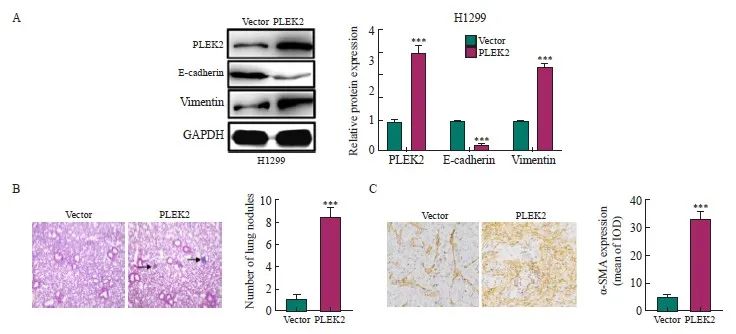
图2 PLEK2过表达促进肺癌细胞EMT
Fig. 2 PLEK2 overexpression promotes EMT in lung cancer cells
A: The expression of PLEK2, E-cadherin and vimentin in H1299 cells transfected with Vector control and PLEK2 overexpression lentivirus was analyzed by protein blot. B: H-E staining of lung tissue and quantitative analysis of metastatic nodules. C: Immunohistochemical analysis of α-SMA in lung tissue metastases. IOD: Integral optical density. ***: P<0.001, compared with Vector group.
2.3 PLEK2过表达上调肺癌细胞及其外泌体中的miR-196a表达
为了探索PLEK2在CAFs激活中高表达的机制,本研究进一步分析了这些细胞来源的外泌体(分别命名为Vector_exo和PLEK2_exo)中的miRNA表达谱。由武汉华大基因科技有限公司完成测序工作,选择分析的miRNA种类是随机的。RNA测序结果表明,与Vector组和Vector_exo组相比,在PLEK2组细胞及其外泌体中miR-196a表达水平最高(表1)。为了证实测序结果,我们进行了RTFQ-PCR分析,发现与Vector组相比, PLEK2组H1229细胞中miR-196a表达水平显著增加(P<0.05),并且PLEK2_exo中miR-196a表达水平显著高于Vector_exo(P<0.05,图3)。
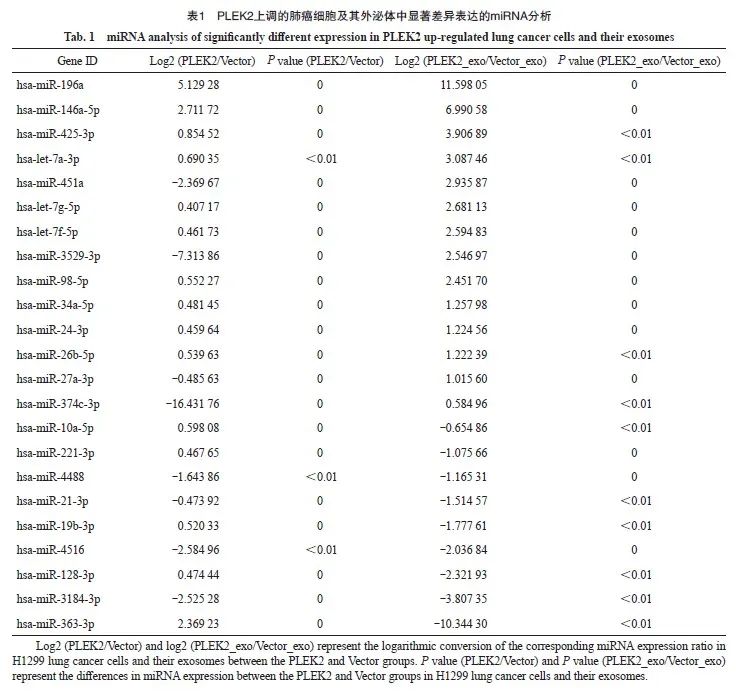
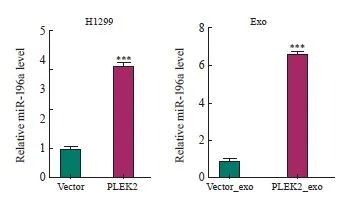
图3 PLEK2过表达上调肺癌细胞及其外泌体中的miR-196a表达
Fig. 3 PLEK2 overexpression upregulates miR-196a expression in lung cancer cells and their exosomes
***: P<0.001, compared with the Vector group or Vector_exo group.
2.4 肿瘤细胞来源的外泌体miR-196a被成纤维细胞摄取并促进CAFs活化
本研究假设肿瘤来源的外泌体miRNA通过激活成纤维细胞驱动H1299细胞的EMT而增强肺癌转移。为了证明这一假设,本研究首先从Vector组和PLEK2组H1299细胞中分离出外泌体。这些外泌体呈现双层杯状形态,直径为100~150 nm(图4A)。Western blot分析显示,外泌体中的外泌体标志物CD9、CD63和CD81阳性表达,但ER膜标志物calnexin阴性表达(图4B)。为了追踪外泌体的递送,本研究用膜染料PKH26标记了H1299细胞来源的外泌体。结果,在共培养48 h后,标记的外泌体被递送至MRC-5细胞(图4C)。
将Vector_exo、PLEK2_exo分别与MRC-5细胞一起温育。与Vector_exo组相比,PLEK2_exo组MRC-5细胞中miR-196a、α-SMA和FAP表达水平显著增加(P<0.05,图4D)。为了探究外泌体对成纤维细胞活化的影响是否是由于miR-196a的转移,用miR-196a模拟物直接转染MRC-5细胞。与NC相比,miR-196a转染的MRC-5细胞中α-SMA和FAP表达水平显著增加(P<0.05,图4E、4F)。相反,通过miR-196a抑制剂(si-miR-196a#1和si-miR-196a#)转染,α-SMA和FAP表达水平被显著抑制(P<0.05,图4G、4H)。
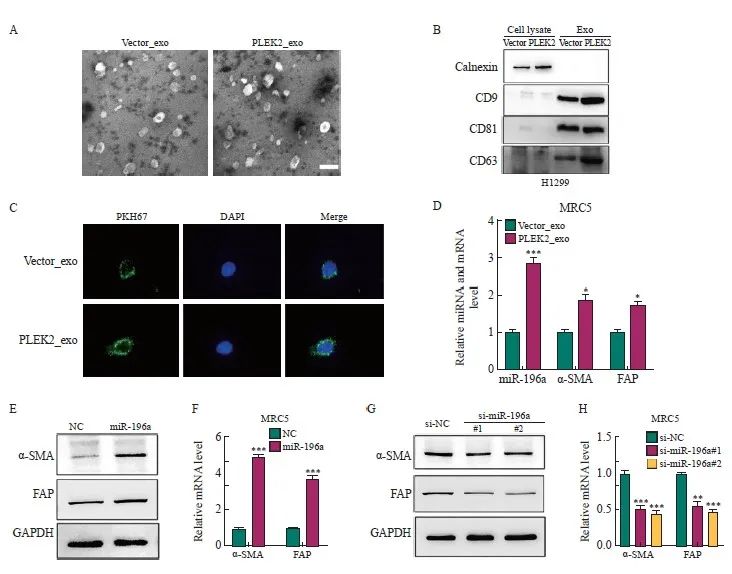
图4 肿瘤细胞来源的外泌体miR-196a被成纤维细胞摄取并促进CAFs活化
Fig. 4 Exosomes miR-196a derived from tumor cells are taken up by fibroblasts and promote the activation of CAFs
A: Transmission electron micrographs of Vector_exo and PLEK2_exo (scale=100 nm). B: Western blot analysis of the expression of CD9, CD63, CD81 and calnexin in exosomes. C: After co-culture for 48 h, the internalization of Vector_exo and PLEK2_exo labeled with PKH26 by MRC-5 cells was examined by confocal microscope. D: RT-qPCR analysis of miR-196a, α-SMA, FAP in MRC-5 cells treated with Vector_exo, PLEK2_exo. E, F: Western blot and RT-qPCR were used to determine the protein and mRNA levels of α-SMA and FAP in MRC-5 cells transfected with miR-196a mimetic (miR-196a) and NC. G, H: Western blot and RT-qPCR were used to determine the protein and mRNA levels of α-SMA and FAP in MRC-5 cells transfected with miR-196a inhibitors (si-miR-196a#1 and si-miR-196a#) and control (si-NC). *: P<0.05, compared with the Vector_exo group; **: P<0.01, compared with the Vector_exo group or si-NC group; ***: P<0.001, compared with the Vector_exo group, NC group or si-NC group.
2.5 CAFs的激活促进肺癌细胞侵袭和转移
为探究由外泌体miR-196a激活的成纤维细胞是否能促进肺癌细胞的侵袭和转移,将用miR-196a模拟物及对照转染的MRC-5细胞的条件培养基(miR-196a-CM、NC-CM)或用Vector_exo、PLEK2_exo处理MRC-5细胞的条件培养基(Vector_exo-CM、PLEK2_exo-CM)处理H1299细胞。结果显示,与NC-CM组相比,miR-196a-CM组H1299细胞的转移[(156±8) vs (324±12)]、侵袭[(84±5) vs (207±10)]细胞数和vimentin表达[(1.00±0.02) vs (2.26±0.14)]均显著增加(P<0.001),E-cadherin表达[(1.00±0.02) vs (0.28±0.02)]显著降低(P<0.001,图5A、5B)。此外,与Vector_exo-CM组相比,PLEK2_exo-CM组H1299细胞的转移[(192±10) vs (403±22)]、侵袭[(11 4 ±5 ) vs ( 287±18)]细胞数和vimentin表达[(1.00±0.02) vs (2.15±0.13)]均显著增加(P<0.01),E-cadherin表达[(1.00±0.02) vs (0.36±0.03)]显著降低(P<0.001,图5C、5D)。
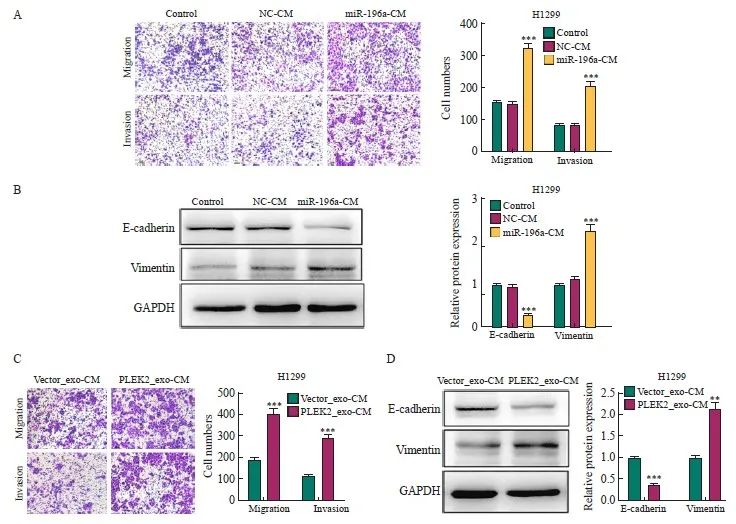
图5 CAFs激活促进肺癌的侵袭和转移
Fig. 5 CAFs activation promotes the invasion and metastasis of lung cancer
A: The metastatic and invasive ability of H1299 cells treated with miR-196a-CM was determined by transwell test. B: the expression level of EMT-related proteins in H1299 cells treated with miR-196a-CM. C: The metastatic and invasive ability of H1299 cells co-cultured with Vector_exo-CM and PLEK2_exo-CM was determined by transwell test. D: The expression level of EMT-related proteins in H1299 cells co-cultured with Vector_exo-CM and PLEK2_exo-CM. **: P<0.01, compared with Vector_exo-CM group; ***: P<0.001, compared with NC-CM group or Vector_exo-CM group.
3 讨 论
在TME中,成纤维细胞响应来自癌细胞的信号而被激活,获得α-SMA阳性的CAFs表型[10]。尽管CAFs在不同人群中存在显著的表型异质性,但CAFs通过ECM的重塑和血管生成因子的分泌促进恶性细胞的生长和迁移[11]。因此,CAFs已被用作抑制转移的治疗靶点。然而,阐明介导CAFs和肿瘤细胞之间通讯的信号分子仍然是一个巨大的挑战,这些信号分子对癌症转移至关重要。
PLEK2是PLEK1的一种膜蛋白和旁系同源物,具有2个PH结构域,其能与磷脂酰肌醇3-激酶产生的膜相关磷脂酰肌醇结合,后者参与调节肌动蛋白细胞骨架组织和细胞扩散[12]。越来越多的证据[13-14]表明,PLEK2在多种肿瘤中高表达,并与癌症进展和转移密切相关。在肺癌中,PLEK2通过泛素依赖的SHIP2降解介导非小细胞肺癌的转移和血管侵袭[15]。此外,PLEK2可作为LUAD患者的生存预后指标[16]。本研究通过对公共数据集进行生物信息学分析,发现PLEK2可能是肺癌的潜在分子靶标,且PLEK2过表达的H1299细胞在裸鼠体内成瘤率增强,肿瘤中的α-SMA表达增强,表明PLEK2介导的肺癌细胞和CAFs相互交流促进了肿瘤的恶性发展。最近研究[17]显示,转化生长因子-β(transforming growth factor-β,TGF-β)通过诱导的PLEK2上调促进食管鳞状细胞癌的转移和耐药。值得注意的是,在TME中,CAFs是TGF-β的主要来源,这可能是PLEK2在肺癌细胞中高表达的主要原因。
CAFs是肿瘤组织中活化的成纤维细胞,通过产生各种类型的细胞因子促进恶性进展、侵袭和转移[18]。在TME中,基质CAFs和癌细胞通过多种直接和间接机制相互作用,其中外泌体miRNA可以作为关键介质发挥作用[19],如肝细胞癌来源的外泌体miR-21通过PI3K信号促进CAFs的激活[20]。本研究发现miR-196a在PLEK2过度表达的肺癌细胞的外泌体中的表达显著上调。miR-196a在不同类型的癌症中作为致癌miRNA发挥作用[21]。在非小细胞肺癌中,miR-196a是一种肿瘤启动子,可增加癌症细胞的增殖、侵袭能力和耐药性[22]。此外,有研究[23]显示,来源于CAFs的miR-196a通过外泌体转移到头部和颈部癌症细胞中,以增加癌细胞增殖、存活能力和耐药性。这些前期结果限制性地描述了miR-196a的作用,因为上述研究仅关注癌细胞。在本研究中,miR-196a过表达刺激MRC-5细胞表达肌成纤维细胞标志物,如α-SMA和FAP,并通过条件培养基促进肺癌细胞侵袭和转移。此外,H1299细胞来源的PLEK2_exo又能促进MRC-5细胞发生EMT。因此,外泌体miR-196a在PLEK2过表达的癌细胞和基质CAFs之间的相互作用中发挥重要作用,形成了促进肺癌细胞转移的反馈环[24]。
综上所述,PLEK2的上调能够增强肺癌细胞来源的外泌体miR-196a水平,从而促进CAFs的激活。激活的CAFs能够进一步增强肺癌细胞的侵袭能力。因此,通过miR-196a抑制剂阻断外泌体miRNA介导的CAFs激活可能作为抑制PLEK2诱导的肺癌转移的有前景的治疗策略。此外,最近研究[25]显示,PLEK2抑制剂在抑制骨髓增生性肿瘤增殖方面展现出显著的治疗效果。因此,miR-196a抑制剂和PLEK2阻断剂或常规抗癌药物的联合治疗可能是同时靶向TME中的癌症细胞和基质细胞的治疗选择。
利益冲突声明:所有作者均声明不存在利益冲突。
作者贡献声明:
王蔓莉:进行实验并撰写文章;
陈辉:实验设计,审查文章;
段智:进行实验;
许奇美,李贞:修改文章。
[参考文献]
[1] SUNG H, FERLAY J, SIEGEL R L, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249.
[2] MILLER K D, NOGUEIRA L, DEVASIA T, et al. Cancer treatment and survivorship statistics, 2022[J]. CA Cancer J Clin, 2022, 72(5): 409-436.
[3] CHAFT J E, RIMNER A, WEDER W, et al. Evolution of systemic therapy for stagesⅠ-Ⅲ non-metastatic non-smallcell lung cancer[J]. Nat Rev Clin Oncol, 2021, 18(9): 547-557.
[4] ENTEZARI M, GHANBARIRAD M, TAHERIAZAM A, et al. Long non-coding RNAs and exosomal lncRNAs: potential functions in lung cancer progression, drug resistance and tumor microenvironment remodeling[J]. Biomedecine Pharmacother, 2022, 150: 112963.
[5] WANG G C, ZHOU Q, XU Y, et al. Emerging roles of pleckstrin-2 beyond cell spreading[J]. Front Cell Dev Biol, 2021, 9: 768238.
[6] WEN Y S, GUO G R, YANG L J, et al . Atumor microenvironment gene set-based prognostic signature for non-small-cell lung cancer[J]. Front Mol Biosci, 2022, 9: 849108.
[7] HU S C, MA J H, SU C, et al. Engineered exosome-like nanovesicles suppress tumor growth by reprogramming tumor microenvironment and promoting tumor ferroptosis[J]. Acta Biomater, 2021, 135: 567-581.
[8] PATHANIA A S, PRATHIPATI P, CHALLAGUNDLA K B. New insights into exosome mediated tumor-immune escape: clinical perspectives and therapeutic strategies[J]. Biochim Biophys Acta Rev Cancer, 2021, 1876(2): 188624.
[9] NIU L R, YANG W L, DUAN L L, et al. Biological implications and clinical potential of metastasis-related miRNA in colorectal cancer[J]. Mol Ther Nucleic Acids, 2021, 23: 42-54.
[10] TAN Z F, XUE H B, SUN Y L, et al. The role of tumor inflammatory microenvironment in lung cancer[J]. Front Pharmacol, 2021, 12: 688625.
[11]XU K H, ZHANG C P, DU T T, et al. Progress of exosomes in the diagnosis and treatment of lung cancer[J]. Biomedecine Pharmacother, 2021, 134: 111111.
[12]ZHAO X Y, SHU D L, SUN W J, et al. PLEK2 promotes cancer stemness and tumorigenesis of head and neck squamous cell carcinoma via the c-Myc-mediated positive feedback loop[J]. Cancer Commun, 2022, 42(10): 987-1007.
[13]LIU J H, CHEN H, QIAO G B, et al. PLEK2 and IFI6, representing mesenchymal and immune-suppressive microenvironment, predicts resistance to neoadjuvant immunotherapy in esophageal squamous cell carcinoma[J]. Cancer Immunol Immunother, 2023, 72(4): 881-893.
[14]SHAO W, AZAM Z, GUO J T, et al. Oncogenic potential of PIK3CD in glioblastoma is exerted through cytoskeletal proteins PAK3 and PLEK2[J]. Lab Invest, 2022, 102(12): 1314-1322.
[15]WU D M, DENG S H, ZHOU J, et al. PLEK2 mediates metastasis and vascular invasion via the ubiquitin-dependent degradation of SHIP2 in non-small cell lung cancer[J]. Int J Cancer, 2020, 146(9): 2563-2575.
[16]XIA Q L, HE X M, MA Y, et al. 5-mRNA-based prognostic signature of survival in lung adenocarcinoma[J]. World J Clin Oncol, 2023, 14(1): 27-39.
[17]WANG F, ZHANG C Q, CHENG H, et al. TGF-β-induced PLEK2 promotes metastasis and chemoresistance in oesophageal squamous cell carcinoma by regulating LCN2[J]. Cell Death Dis, 2021, 12(10): 901.
[18]WU C X, GU J M, GU H B, et al. The recent advances of cancer associated fibroblasts in cancer progression and therapy[J]. Front Oncol, 2022, 12: 1008843.
[19]SHELTON M, ANENE C A, NSENGIMANA J, et al. The role of CAF derived exosomal microRNAs in the tumour microenvironment of melanoma[J]. Biochim Biophys Acta Rev Cancer, 2021, 1875(1): 188456.
[20]MA Y N, WANG S S, LIEBE R, et al. Crosstalk between hepatic stellate cells and tumor cells in the development of hepatocellular carcinoma[J]. Chin Med J, 2021, 134(21): 2544-2546.
[21]CUI J Z, YUAN Y, SHANMUGAM M K, et al. MicroRNA-196a promotes renal cancer cell migration and invasion by targeting BRAM1 to regulate SMAD and MAPK signaling pathways[J]. Int J Biol Sci, 2021, 17(15): 4254-4270.
[22]QIU H, XIE Z Q, TANG W F, et al. Association between microRNA-146a, -499a and-196a-2 SNPs and non-small cell lung cancer: a case-control study involving 2249 subjects[J]. Biosci Rep, 2021, 41(2): BSR20201158.
[23]QIN X, GUO H Y, WANG X N, et al. Exosomal miR-196a derived from cancer-associated fibroblasts confers cisplatin resistance in head and neck cancer through targeting CDKN1B and ING5[J]. Genome Biol, 2019, 20(1): 12.
[24]LIU E C, LEE T T, WU S L, et al. Abstract 1035: PLEK2 promotes epithelial mesenchymal transition through increasing Snail1 in lung cancer cells[J]. Cancer Res, 2019, 79(13_Supplement): 1035.
[25]HAN X, MEI Y, MISHRA R K, et al. Targeting pleckstrin-2/Akt signaling reduces proliferation in myeloproliferative neoplasm models[J]. J Clin Invest, 2023, 133(6): e159638.


